Condorchem Envitech Offer
At Condorchem Envitech, we have an extensive experience in the design of zero liquid discharge (ZLD) plants for the treatment and reuse of industrial wastewater. Every project is customized to meet the specific goals and needs of the customer, taking into consideration different variables, such as the characterization of the effluent, the volume of wastewater to be treated, the sources of energy available, or the ROI of the project.
Condorchem Envitech has a solid team of experts, backed by hundreds of successfully executed projects. We have carried out more than 400 wastewater evaporation and crystallization projects around the globe, some of which include a zero liquid discharge system.
It is important to highlight that vacuum evaporators and crystallizers are currently the best available technologies for the effective implementation of a ZLD system.
Zero liquid discharge systems
A zero liquid discharge system is the best way to guarantee that the activity of a company will not have a significant environmental impact, thanks to a reduction on the use of natural resources and the minimization of the waste that is generated.
Furthermore, zero liquid discharge (ZLD) is often synonymous of circular economy and costs reduction, as the amount of waste to be managed and the water consumption are significantly reduced, if not completely erased. On the other hand, raw materials and by-products can be recovered for it reuse or commercialization.
At Condorchem Envitech, we are experts in the design, manufacturing and installation of vacuum evaporation plants and crystallization systems, the best available technologies today to obtain a zero liquid discharge.
Through a custom design and the proper combination of the different available technologies, especially evaporators, membranes and crystallizers, we have helped many of our clients to reach this goal.
As mentioned above, the main available technologies to for a zero liquid discharge system are:
Thus, microfiltration and ultrafiltration units are commonly used as pre-treatment, whereas processes such as reverse osmosis are used as a primary treatment to recover around 80% of the water, while concentrating the contaminants in a rejected liquid stream. This is followed by thermal separation processes, such as vacuum evaporation and crystallization, which focus on concentrating the reject from membrane technologies, producing distilled water that can be reused in the production process, and dry solid residues, which can sometimes be reused as raw materials, by-products, source of energy, etc.
Vacuum evaporation is a reliable, durable and efficient process that can produce up to 99% of clean water while minimizing the final amount of waste thanks to its great concentration capacity. This is the reason why evaporation and crystallization are essential in all zero liquid discharge systems.
Technologies for zero liquid discharge
As mentioned before, vacuum Evaporation is the core technology in a ZLD system. Vacuum evaporators are know for its efficiency and versatility in the minimization of industrial wastewater and other liquid waste.
Some of the most important types of vacuum evaporators are the following:
- Heat pump vacuum evaporators
- Mechanical vapor compression vacuum evaporators
- Multiple-effect vacuum evaporators powered by hot water
The goal of a ZLD plant is to avoid the discharge of effluents into the environment and to promote the reuse of water and other resources, but what happens when this is not possible? If the final waste still needs to be managed externally, it should be minimized as much as possible and there are some technologies that can make a big difference in sludge minimization:
- Crystallization
- Thermal drying
- Liquid stabilization
Crystallization
Crystallization generates solid crystals that are separated from a solvent (normally water).
Industrial crystallization essentially consists of temporarily obtaining super saturation of a solute above equilibrium, which is the driving force of the process.
This can be achieved by re-concentrating the solute by evaporation of a solvent, cooling the solution or by action of the other chemical product that is added to the solution to decrease the solubility of the original solute, or even a combination of all three processes.
Crystallization also verifies different substantial stages that are distinguished from super saturation in addition to marking the kinetics of crystal formation and their size. Acting on temperature, agitation and time, it is possible to obtain very thin or thick crystals using this pattern.
Evaporative crystallizers work by use of a vacuum, evaporating water at a reduced temperature (35-80 ° C). Water is condensed and reused as distilled water. The evaporation vessel is configured with a heating jacket system, where the heating fluid (steam, hot water, thermal fluid) circulates. This special configuration achieves high concentrations in the chamber with the presence of solids without representing a problem for the process.
The outlet of the crystallizer normally requires the help of some final system for salt dehydration.
- Centrifuge: This unit facilities batch dehydration of large quantities of crystals of all types of salts.
- Drying Filter: the batch of mother liquor and salts is poured over a filter that drains the liquid that returns to the evapo-crystallizer header. Meanwhile, the salts are retained and separated by a traveling scraper that deposits them into a container.
- Drainage Container: Follows the same procedure as above but its larger dimensions can treat greater quantities of crystallized salts.
- Rotating Drum: Outer cylinder has a cooling jacket and a scraper that removes crystals that are deposited on the inner surface. The liquid to be crystalized comes from a concentrated phase of evaporation and is therefore hot. The cooling fluid can be water from a refrigeration circuit with an evaporation tower or refrigerated fluid that is kept at a very low temperature by means of industrial cooling equipment.
- Decanting Reactor: This process utilizes previous evaporation to concentrate the solute but in the equilibration zone. Then, a dosage of a chemical specifically studied for each use, it may be another salt, a solvent, a polymer, etc. An imbalance occurs in original solution leading to the precipitation of crystals that are extracted from the reaction tanks by a specifically designed device. This process allows fractional crystallization and obtains different crystals separated from substances of high added value.
Spray Drying
Spray drying consists of spraying a solution rich in dissolved solids, not in suspension, in a chamber that is kept warm by the action of flue gases from a burner or hot air (180 to 400 °C).
Upon contact with the temperature, the solvent evaporates instantaneously and the solid precipitates in the bottom of the chamber. A venturi system permits the extraction of the dried solid and it’s separated from water vapor and cold combustion gases (approx. 100 °C) that are emitted to the outside.
A filtering / washing process of the gases controls emissions into the atmosphere. Because spray drying is a process that consumes a large amount of energy (kwt / liter evaporated), it is preferably used after an evaporation process to re-concentrate the solute and decrease the volume of water to evaporate.
The solid obtained can be reused when possible or disposed in a controlled landfill.
Stabilization / Inerting
The stabilization of liquids is highly recommended when liquid waste management is very costly or impossible and when crystallization or spray drying cannot be utilized for technical or investment reasons.
Stabilization consists of mixing liquid waste or pasty residue, previously concentrated by an evaporator, with an inert, low cost material. Normally used for this purpose are clays, quicklime, slaked lime, cement, etc.
Some dehydrating polymers such as bentonite and sepiolite are commonly used. In some cases, other solid waste can be utilized (e.g., sewage sludge, ash, slag, etc.).
The mixing process is done in batches or in continuous operation in a unit named BLENDER, which consists of a drum where the feeding liquid or slurry and the solid product stabilizer arrive separately. They are mixed to form a homogeneous mass and are discharged through the front opening to a container.
After a few hours the mix cements. As time passes it loses practically all its humidity, becoming solidified and inert. This product can be taken to a landfill without problem because it will never again dissolve.
The quantity of stabilized cementitious product per liter of liquid or paste depends on the type of residue but is normally between 0.8 and 2 liters of binder per liter of liquid waste or paste.
Our zero liquid discharge technologies
 ENVIDEST MVR FCVacuum evaporators by mechanical vapour recompression and forced circulation
ENVIDEST MVR FCVacuum evaporators by mechanical vapour recompression and forced circulation ENVIDEST MFE 1Single effect thermal vacuum evaporators by forced circulation
ENVIDEST MFE 1Single effect thermal vacuum evaporators by forced circulation ENVIDEST MFE 2Double effect thermal vacuum evaporators by forced circulation
ENVIDEST MFE 2Double effect thermal vacuum evaporators by forced circulation ENVIDEST MFE 3Triple effect thermal vacuum evaporators by forced circulation
ENVIDEST MFE 3Triple effect thermal vacuum evaporators by forced circulation ENVIDEST MVR FF 30 – 2500Falling film vacuum evaporators by mechanical vapour recompression and forced circulation
ENVIDEST MVR FF 30 – 2500Falling film vacuum evaporators by mechanical vapour recompression and forced circulation ENVIDEST DPM 1Single effect thermal vacuum evaporators
ENVIDEST DPM 1Single effect thermal vacuum evaporators ENVIDEST DPM 2Double effect thermal vacuum evaporators
ENVIDEST DPM 2Double effect thermal vacuum evaporators ENVIDEST DPM 3Third effect thermal vacuum evaporators
ENVIDEST DPM 3Third effect thermal vacuum evaporators ENVIDEST LT VSHeat pump vacuum evaporators
ENVIDEST LT VSHeat pump vacuum evaporators ENVIDEST EAAtmospheric evaporators
ENVIDEST EAAtmospheric evaporators DESALT LT DRYHeat pump vacuum crystallizers
DESALT LT DRYHeat pump vacuum crystallizers DESALT LT VRHeat pump vacuum crystallizers
DESALT LT VRHeat pump vacuum crystallizers DESALT MFEThermal vacuum crystallizers by forced circulation
DESALT MFEThermal vacuum crystallizers by forced circulation DESALT VRThermal vacuum crystallizers
DESALT VRThermal vacuum crystallizers DESALT MVR FCVacuum crystallizers by mechanical vapour recompression and forced circulation
DESALT MVR FCVacuum crystallizers by mechanical vapour recompression and forced circulation DESALT DRYThermal vacuum crystallizers
DESALT DRYThermal vacuum crystallizersBenefits of a ZLD system
Zero liquid discharge is the most sustainable, and often the most cost-effective, alternative that industries can adopt for wastewater management. Zero liquid discharge can be achieved by implementing technologies and processes that enable the reuse of liquid effluents with a dual purpose in mind: first, reducing water consumption as much as possible and, secondly, minimizing the volume of waste to be externally managed.
Thus, a zero liquid discharge plant concentrates pollutants by thermal treatment and, finally, the effluents are reduced to a dry residue, while the extracted water can be reused again in the process.
The main advantagentages of the implementation of a zero discharge system are the following:
- Reduction in the company’s environmental impact by minimizing the production of liquid waste, sludge and solid waste.
- Savings in external waste management.
- Reduction in the consumption of water by reusing the clean water produced in the ZLD plant. This advantage also results in an even greater reduction of the company’s environmental impact.
- Savings in water cost.
- Potential savings in raw materials
- Possible increase in the income due to the commercialization of by-products
- Improve the company’s environmental responsibility and its commitment to sustainability.
- Compliance with the most demanding environmental regulations.
- Savings in the taxes applied to waste disposal or discharge.
- Avoid potential sanctions for not complying with the environmental regulations in force.
- Increased flexibility in the management of liquid effluents produced by not depending on any external agent.
- Easy to operate.
- High degree of automation.
- No need for specialized technical personnel.
- Savings in the consumption of chemical reagents.
- A high degree of self-sufficiency with regard to water consumption.
- Possibility of reusing secondary heat from other processes, dramatically reducing operating costs.
Concept
An optimized ZLD system is the best option to recover high quality water and valuable materials from wastewater.
A zero liquid discharge system focuses on the use of techniques and processes to transform a polluted effluent into valuable resources and achieve the following key targets:
- Reduce the consumption of water taken from outside the company as far as possible
- Reuse of water in the production process.
- Reuse or sell raw materials and by-products for other industries and applications.
- Correctly treat polluted effluents to prevent environmental pollution.
- Minimize the quantity of waste generated.
- Use the concentrated waste as an alternative fuel in cogeneration equipment or other systems.
Installing a ZLD system has many advantatges if we compare it with consuming as much water as necessary and discharging the effluents produced are numerous. The following are the most important:
- A dramatic saving in the consumption of water as the majority of water is reused.
- Reduction of the company's footprint as no waste is discharged.
- Minimal waste production with its inherent environmental and economic benefits.
- Reduction of economic costs.
- A high degree of self-sufficiency regarding water consumption.
Installing a ZLD plant
When possible, the implementation of a ZLD plant is highly advisable, as it is the best solution to apply the principles of circular economy and responsible use of natural resources.
Zero liquid discharge systems are flexible and scalable, as they can be customized to manage any kind of liquid waste. The only case where they can present some complexity in their design is when the wastewater produces volatile organic compounds. One of the advantages of a ZLD plant is its versatility to manage a wide range of pollutants and its vast scope of applications.
The main obstacle when installing a zero liquid discharge system may be its cost, as it is not always the cheapest solution. To check if a zero discharge system is efficient and cost-effective, its CAPEX and OPEX need to be analyzed before any decision is made.
Although conventional technologies can be technically feasible in a large number of cases, there are situations where these traditional systems are not a cost-effective option. This happens when the wastewater to be managed presents a complex characterization and the pollutants can not be removed without vacuum concentration technologies, which are the core of a zero liquid discharge plant.
In other cases, the treated effluent cannot be discharged due to geographical reasons (water scarcity) or because it requires a high investment to meet the increasingly stringent environmental regulations. In these situations, a zero discharge system must be evaluated.
Sectors and applications
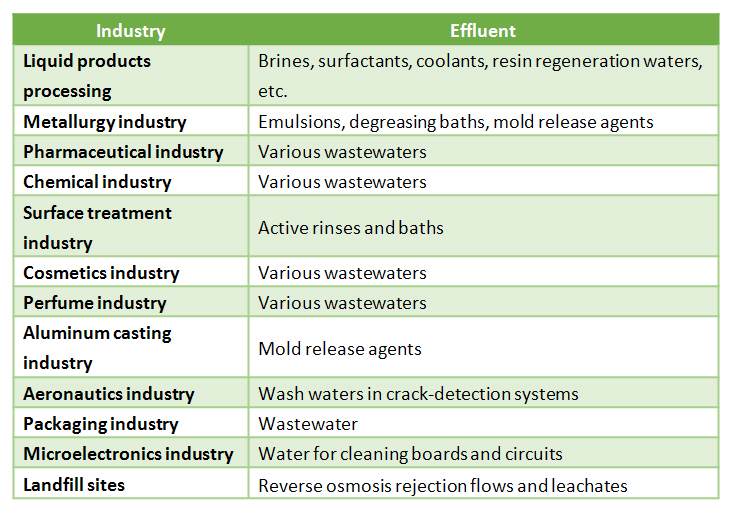
The implementation of zero liquid discharge systems is most common in the following sectors:
- Food industry
- Metals industry (e.g. emulsions, degreasing baths and release agents)
- Pharmaceutical industry
- Chemical industry
- Surface treatment industry
- Cosmetics industry
- Perfume industry
- Aluminum casting industry
- Aeronautics industry
- Microelectronics industry
- Municipal solid waste dumps
- The preparation of pickles, preserves and salted fish.
- Emulsions and degreasing baths in the metal-mechanical industry.
- Equipment washes in the chemical, pharmaceutical, cosmetic and perfumery industries.
- Leachate from municipal solid waste dumps.
- Demolding agents in the aluminum injection industry.
- Washes from the construction of plates and electronic circuits.
Although these are the most common sectors, the number of possible applications of a ZLD plant is as broad as the number of different activities that generate liquid effluents, with some minor limitations.
The activities in which a zero waste management system is particularly useful and advantageous are summarized in the following chart.
Figure 2. Industrial sectors in which a zero waste system is a key tool.
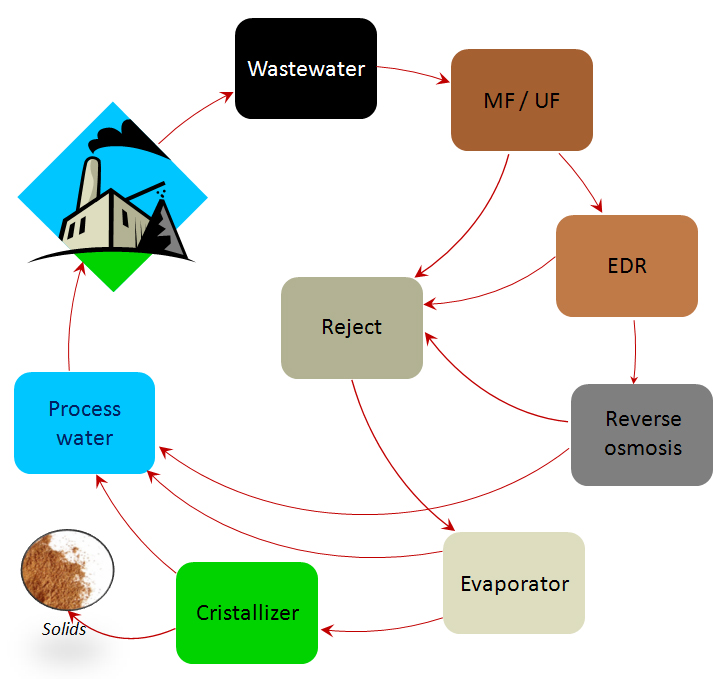
The virtuous circle representing the zero waste concept is shown in figure 3. Not all the processes shown are required in all cases.
Membrane processes (MF, UF, RED, and RO) generate a large volume of water suitable for reuse.
And thermal processes (evaporation and crystallization) treat the rejection flows produced by the former processes, producing yet more water suitable for reuse and a final dry solid residue.
This diagram shows the typical design of a ZLD system.
ZLD to help prevent water scarcity
Water is a precious resource to be preserved. Water scarcity is a problem common to all continents.
According to the United Nations (Coping with water scarcity. Challenge of the twenty-first century. UN-Water, FAO, 2007), water scarcity affects 1200 million people–almost a fifth of the world’s population–living in regions with physical water scarcity, with a further 500 million people close to this situation.
Water scarcity is one of the most important problems that will be faced by many societies in the 21st century.
Although there is sufficient water on the planet to supply 7000 million people, it is not equally distributed, and the majority is squandered, polluted or managed unsustainably.
Hydrology experts assess water scarcity on the basis of the ratio between the population and the water available.
A region is considered to suffer from hydric stress when the annual quantity of water available is less than 1700 m3 per person.
When this ratio reaches 1000 m3 per person, the situation is one of water scarcity. A figure of less than 500 m3 per person indicates absolute scarcity.
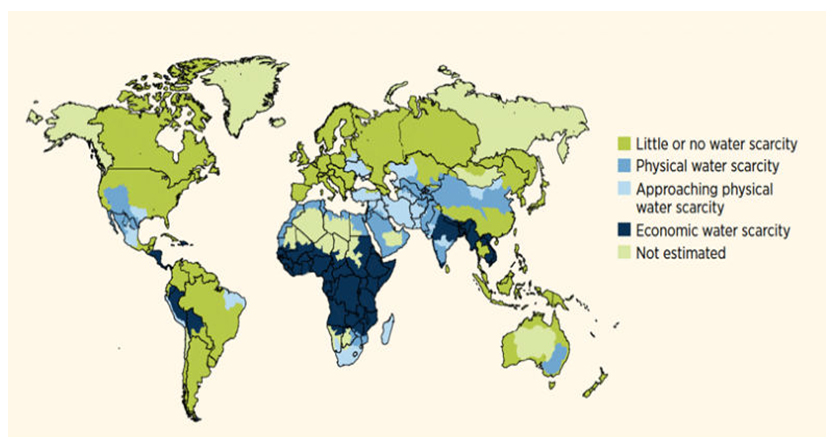
The regions of the world suffering from water scarcity are shown in Figure 1.
A difference can be made between physical scarcity, when the water available is less than the demand, and economic scarcity, in which human, institutional and financial factors limit the access to water despite sufficient water being available.
Figure 1. Global physical and economic water scarcity. Source: World Water Development Report 4. World Water Assessment Program (WWAP), March 2012.
The management of water scarcity does not just involve major construction projects, such as interbasin transfers, to increase the availability of water resources; it is also essential to make better use of the water available without wasting or polluting it.
Water reuse, especially by industries that require large amounts, is essential in order to move towards a solution to this problem. This is why increasing efforts are being devoted today to maximize the removal or reuse of waste.