Industrial wet scrubbers for air pollution control
At Condorchem Envitech we design and manufacture scrubber systems, which effectively remove pollutants from contaminated industrial air emissions.
Our wet and dry scrubbers can be used effectively for the treatment of particles, toxic pollutants and inorganic fumes, vapors and gases, such as: chromic acid, hydrogen sulfide, ammonia, chlorides, fluorides and SO2.
Our scrubber systems are designed to provide a lower pressure drop, which translates into lower energy consumption.
In our scrubbers the removal of pollutants is produced by a chemical reaction between the air and the purification solution (the chemical agent) contained in the purification tower. Pollutants are transferred by absorption from air to water. This technology can operate in two different ways:
1. By inertial impact through reaction with a specific chemical agent that varies according to the type of pollutant to be eliminated.
2. By physical absorption carried out by a solvent in the liquid phase.
Each wet scrubber is designed and customized based on the specific needs of each client and the specific regulations that must be met.
Condorchem Envitech designs a wide variety of scrubbers (traditional, bioscrubbers, Venturi scrubbers, quenchers). Each type of scrubber offers specific advantages and can be classified based on various criteria:
- Perforated plate columns
- Traditional and special filling columns in co-current or countercurrent
- Spray wash columns
Filling column
Setting
The configuration depends on the space available at the customer's premises. Depending on the available space, the following configurations can be installed:
- Horizontal
- Vertical
Material
The decision of the manufacturing material is mainly determined by the temperature that is reached inside the scrubber:
- Plastic: The most common options are polypropylene or PVDF. These equipments are designed up to temperatures of 80-90ºC
- Steel: It can be made of carbon steel, AISI 304 or AISI 316. These equipments are installed when working at high temperatures or for pressure equipment (PED). Steel can be internally coated with a high chemical resistant epoxy paint / resin if the stream carries a lot of acidic contaminants.
Wash solution
The equipment can be designed with one, two or three washing stages. The 3SCR is a three stage wash removal system for high contaminant streams.
- Acid: For abatement of alkaline pollutants, mainly ammonia. Commonly used solutions are sulfuric acid and hydrochloric acid.
- Basic: For abatement of acid pollutants, such as sulfuric, hydrochloric, nitric, hydrofluoric or hydrobromic acid. Sodium hydroxide is mainly used as a solution.
- Oxidizer: For odor removal and disinfection. Sodium hypochlorite or peroxides are mainly used.
Our wet scrubbers
Main characteristics
- Lower tank to contain the wash water with service accessories
- Bottom drain and automatic exhaust valve
- Recharge of chemical reagent systems and make-up water (ARRS and AWRS)
- Three-point level control for purge control: reintegration
- Manhole for inspection and maintenance operations
- Wash water ramp
- Centrifugal water circulation pump, vertical or horizontal axis, made of stainless steel or polypropylene
- Control Panel
Optional
- Structure in SS AISI 304, AISI 316, FRP, PVDF, PE
- Fan, variable frequency converter, soundproof booth
- Antifreeze system
Advantages of industrial wet scrubbers
- No limit to treated flow
- Wide variability in the load to be treated
- High efficiency
- Uses the most common reagents
- Fully automated to minimize maintenance cost
Applications of industrial scrubbers
The technique is applicable to a wide variety of industrial emissions, of which the following are the most common:
- Wastewater treatment plants: Pumping wells, drainage wells, thickeners, pre-treatment and sludge dehydration.
- Composting plants
- Food industry: smells from slaughterhouses, animal fat treatment, fish processing plants
- The chemical and pharmaceutical industry: gases from reactor vents
- Incineration and blanketing gases
Introduction to industrial wet scrubbers
Gas scrubbing is the technology by which cleans a gaseous emission of the contaminants it contains. Typically, the air pollutant molecules are separated from the gaseous stream upon contact with a liquid, which may be water, a chemical reagent or a combination of these. The gaseous flow, once washed, is free of contaminants and can be emitted into the atmosphere. Contact of the contaminant with the liquid depends on the type of washer, which may be by wet packing, bubbling or aerosol, for example.
Generally, gas scrubbing is used to remove air pollutants such as smells, vapors and toxic gases. Therefore, in most cases, the pollutants present in the gases are susceptible to being oxidized or absorbed in an acid or alkaline medium. Thus, species derived from nitrogen can be absorbed in an acid environment, while species derived from sulfur are sensitive to absorption in an alkaline or oxidizing medium. In some cases, the contaminants are very soluble in water, so no chemical reagent is needed.
Types of filling columns
Whatever the type of column that best suits the particular conditions of each customer to ensure the success of the scrubbing system, the extensive experience of the Condorchem Envitech technical team has demonstrated that a particularly critical parameter is the effective design of the contaminated air collection systems, so that the entire gas stream to be treated is efficiently conducted to the gas scrubber. The efficiency of the collection and conduction system is equal in importance to the degradation efficiency of the wash column contaminants. The most effective designs are:
Packed or filled tower
In this gas scrubber design the tower is partially filled with a support with a high specific surface area (e.g. Raschig rings, Pall rings and Berl saddles) and the liquid that will absorb or react with the contaminant. The carrier particles encourage the gas entering through the bottom of the tower to be move upwardly between the interstitial spaces left by the carrier particles, enhancing the absorption efficiency of the liquid. These towers have a high contact efficiency.
Perforated plate tower
In these types of towers, the liquid descends from plate to plate while the gas rises and passes through holes in the plates, thus ensuring effective contact between the two phases. These towers are particularly useful when liquid and gas loads are very variable, or a large diameter column is required, or fouling is likely to occur (plate columns are easier to clean than packed) or thermal or mechanical stresses occur that may damage the fill.
Spray towers
In this type of gas scrubbing tower, a liquid is injected in the form of an aerosol into the gaseous stream and used when high flow rates are to be treated, usually to remove some pollutant from the flue gases inside of the spray tower. As flue gas scrubber their use is preferred when a low-pressure loss is required and if there are particles in the gaseous stream to be treated.
Functioning/operation of wet scrubbers
There are numerous industrial processes in which pollutant gases are produced that must be treated before being emitted into the atmosphere. An effective technique consists of subjecting the gases to a gas-liquid absorption process. The gases to be treated enter through the bottom of one or more absorption or scrubber towers, partially filled with a liquid (e.g. water, acid solution, alkaline solution, sodium hypochlorite or potassium permanganate) or a combination of liquids, which absorb the pollutants present in the gas. The gas leaves the scrubber without the pollutants being present and may be emitted into the atmosphere. Pollutants that are satisfactorily eliminated through a gas-liquid absorption process are SO2 and the NOX of the combustion gases, the hydrogen sulphide from wastewater treatment plant, COV, carbon monoxide, etc.
For the decontamination of the gas to be exhaustive, the system must be designed such as to maximize the transfer of material from the gas phase to the liquid phase:
- The contaminant and the liquid must be compatible; i.e. the solubility of the former in the second must be sufficiently high.
- The contact surface must be large enough for there to be no limitation of transfer of the contaminant to the absorbent liquid.
- The contact of the contaminants present in the gaseous flow with the liquid depends on the type of absorption tower.
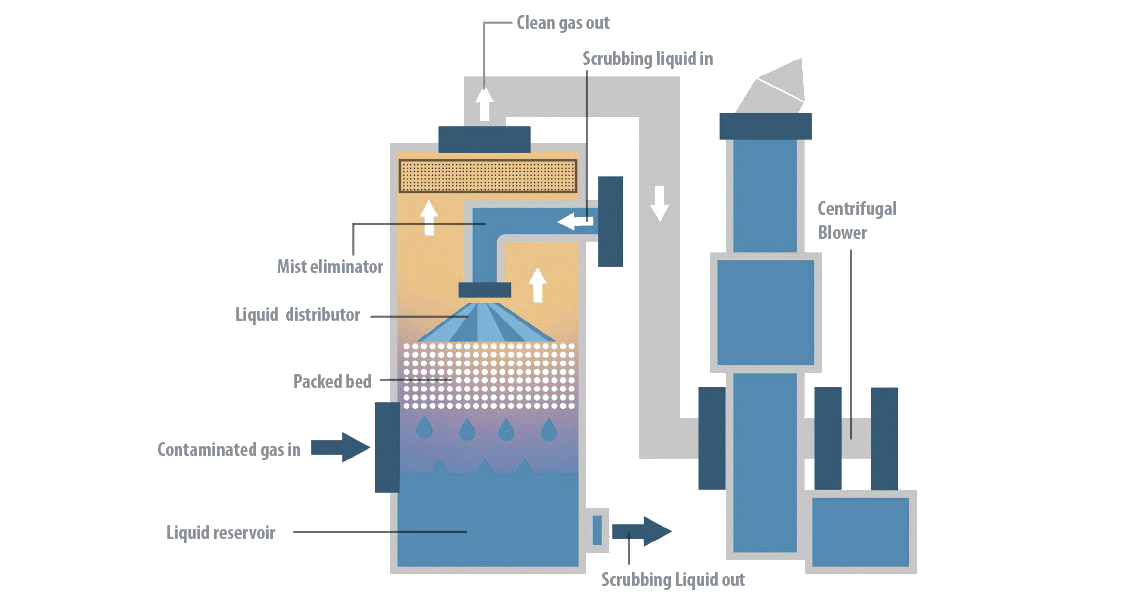
As it passes through the scrubbing tower, polluted air is washed at low speed inside a large contact surface. It is very important to use the correct type of absorbent during this process to ensure greater gas / liquid contact. Once purified, the air goes to the next stage or is released directly into the atmosphere.
1. The air passes through a compact column in a large contact surface.
2. The wash solution is continuously sprayed with the centrifugal pump through the nozzles and automatically replaced as required by ARRS.
3. Automatic water filling system (AWRS) ensures that the working level of the liquid is maintained.
4. The base of the column is a wash solution tank.



