Sections
- Wastewater in mining
- Mining wastewater treatment solutions
- Zero discharge system for saline effluent treatment
- Comparison of zero liquid discharge (zld) with other systems
Mining extraction operations are increasingly affected by strict regulations regarding the wastewater they produce and the need not to dispose of liquid waste that could affect the environment.
These regulations can seriously affect the viability of their businesses if they do not take the necessary measures to treat the wastewater they produce.
Finding the most suitable and sustainable wastewater treatment over time is one of the main challenges a mining company faces in maintaining its operating license.
Condorchem Envitech offers efficient solutions for the mining sector through advanced treatment processes that allow companies to produce clean water for reuse or discharge into the environment without any danger.
WASTEWATER IN MINING
Wastewater can be produced in various ways in a mining operation, depending on whether the mine is underground or open pit, whether it has a positive or negative water balance and depending on the refining process.
Mine water generally has the following components:
- Free acid
- Anions, such as sulfate (SO42-) and carbonate (CO32-)
- Dissolved metals, such as iron (Fe), aluminum (Al), calcium (Ca), magnesium (Mg), manganese (Mn) and sodium (Na).
- Suspended particles
- Chemical products
Wastewater produced by the mining industry is also known as tailings. Tailings contain waste from the extraction processes, equipment washing water and the refining process.
It is a sludge-like waste that contains a mixture of fine gangue particles, chemicals and water; and in many cases is stored in large facilities called tailings dams or ponds.
Tailings represent an important environmental responsibility for a mining operation, since they occupy large areas of land and contain water contaminated with metals, which will eventually need to be treated during operation or after the mine closes down.
MINING WASTEWATER TREATMENT SOLUTIONS
Given the high cost of treating and pumping wastewater produced from a mine, it is important to have processes and technologies that are cost-effective that help prevent uncontrolled discharge of wastewater, resulting in salinization and contamination of surface or groundwater.
There are different technologies available for the treatment of the effluents produced in a mining operation.
Some of these technologies can be combined to achieve the most efficient result, both in environmental and financial terms. The available technologies for the treatment of mining effluents are detailed below:
- Precipitation processes: pretreatment, neutralization, metal removal, chemical desalination
- Membrane technologies: reverse osmosis (RO), nanofiltration (NF), electrodialysis (ED/EDR) and membrane distillation (MD)
- Ion exchange technologies (IX)
- Concentration technologies (vacuum evaporation and crystallization)
- Biological treatment
- Irrigation with impacted mine water
- Freezing technologies (eutectic freeze crystallization, hybrid ice technology)
- Other emerging technologies such as forced osmosis (FO), desalination of carbon nanotubes, biomimetic desalination and nano-electrochemical processes.
- Waste manager
When choosing a solution it is very important to take into account the following factors:
- Estimated volume of wastewater to be treated.
- Characterization of the water to be treated: solids, metals, salts, volatile organic compounds (VOCs), acidity and conductivity, for example.
- Water quality requirements for discharge or reuse.
ZERO DISCHARGE SYSTEM FOR SALINE EFFLUENT TREATMENT
At Condorchem Envitech, we propose a zero liquid discharge (ZLD) solution for the mining industry, as required by law to remove the discharge of chlorides and sulfates in the environment.
Such discharge is a by-product of the mining process, and mitigating its effect on the environment is crucial for the mining sector.
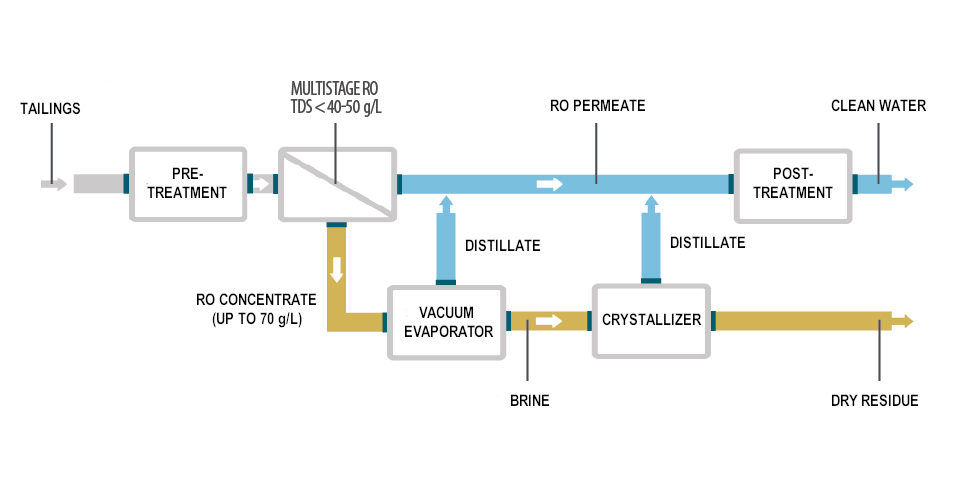
The zero discharge process is carried out by a state-of-the-art membrane treatment that can reach the discharge limits and subsequent treatment by evaporation and crystallization to concentrate the brine from the membrane reject.
Stage 1: Clarification and softening
Wastewater enters aeration tanks that promote the precipitation of dissolved metals, such as manganese and iron. Subsequently, it is transferred to a crystallization tank where chemical softening to reduce alkalinity and hardness occurs. Once rinsed in a sedimentation tank, the mineral sludge is pumped into a drainage system, which may be a press filter or a centrifuge.
Stage 2: Pre-concentration
A 1- or 2-stage reverse osmosis (RO) system together with an electrodialysis reversal (EDR) system perform desalination. The RO system consists of parallel skids, each containing multiple reverse osmosis stages. The EDR treats the concentrate from the RO modules and farther concentrates it up to 15% TDS.
Stage 3: Thermal separation
The reject from stage 2 is a concentrated brine that contains dissolved solids and other components removed from the feed wastewater. The brine is sent to a secondary softening system to remove calcium and magnesium ions before being sent to a heat treatment process consisting of an Envidest® evaporator and a Desalt® crystallizer, both designed by Condorchem Envitech.
The evaporator concentrates the RO+EDR reject and separates most of the water efficiently and cost-effectively. The evaporator and crystallizer are from the MFE (Multiple-Effect Evaporator) series and are heated by waste heat: either hot water or steam from the first effect and cooling water for the final effect.
Another option is the MVR (Mechanical Vapor Recompression) series evaporators and crystallizers that compress the steam created by concentrating the brine and then recycle the steam for use in the heater housing to provide a heat source for the evaporation process.
The choice between a MFE or MVR type evaporator depends on factors such as the relative cost and availability of natural gas and energy.
Meanwhile, the crystallizer can be directly powered with low pressure steam from a natural gas boiler instead of using mechanical vapor recompression.
The brine with a high dissolved solids content produced in the evaporator goes to the crystallizer feed tank and is pumped to the crystallizer for farther concentration.
As the evaporation process continues, concentration of the brine increases until the solution becomes supersaturated and the salts precipitate, resulting in a highly concentrated brine sludge.
The centrifuges dehydrate the brine concentrate to form a solid residue.
This brine treatment and desalination system produces clean water and completely removes liquid waste. The resulting desalinated water can be reused for energy production or discharged.
COMPARISON OF ZERO LIQUID DISCHARGE (ZLD) WITH OTHER SYSTEMS
In most cases, a zero liquid discharge system consists of a combination of technologies, which can be divided into 4 main groups:
- Neutralization
- Desalination
- Evaporation
- Brine / Sludge treatment
The main features and advantages of a zero liquid discharge solution are the following:
1. Zero liquid discharge (ZLD) technologies use evaporation and crystallization as reference processes. These produce salt and water concentrates. This water can be reused in the industrial process or in the aquifer regeneration process itself. The salt concentrate can be crystallized and treated as waste or as a valuable resource.
2. Among the products that can be recovered and recycled through evaporation are lithium, caustic soda and sodium sulfate concentrates.
3. A zero liquid discharge system is a good option where water is scarce, or when environmental regulations require high water quality after the process. These systems can treat large volumes of wastewater.
4. The decrease in the volume of wastewater to be treated reduces waste management costs.
5. A zero liquid discharge system can recycle the wastewater in situ, which has two advantages: (1) the treated water can be reused and (2) liquid waste is not transported in tanks, with the corresponding extra cost this logistical solution implies.
6. Zero liquid discharge (ZLD) technologies require significant energy, although the use of waste heat reduces the cost associated with this. In each case, a detailed analysis of the energy costs and availability has to be done.
7. Zero liquid discharge (ZLD) processes normally have two phases: A pre-concentration stage and an evaporation / crystallization stage. Preconcentration can be done by RO, ED/EDR (electrodialysis), FO (forced osmosis) or MD (membrane distillation).
8. MD (membrane distillation) uses the different vapor pressure between both ends of the membrane allowing heat and mass transfer of the volatile solution components, such as water. It is a relatively simple method that can use waste heat.
9. Pre-concentration is required for good performance in zero liquid discharge processes. Pre-concentration is carried out using the other aforementioned technologies (ED/FO/MD). The pre-concentration phase reduces energy consumption in a zero discharge process.
10. ED (Electrodialysis) techniques are optimal for working at high salinities above 70,000 ppm, since they have a high useful life and their cost is less than that of evaporation / crystallization. Ion blocking is compensated for by changes in polarity. This does not allow for the treatment of microorganisms or organic pollutants. These procedures are limited by a specific charge density, which causes dissociation of the water and is a limitation for the procedure.
11. Forced osmosis (FO) allows the treatment of high salinities with an optimal use of energy, since it can combine thermal energy from waste heat and electrical energy. The disadvantage is that it requires high control of pollutant concentrations and can result in concentration polarization problems.
12. Membrane distillation (MD) processes can separate at lower pressures and temperatures than other techniques, and can use waste heat. This technique has fewer flow limitations caused by concentration polarization. As disadvantages, forced osmosis has high modular costs; surfactants can cause wetting problems in the membrane and lead to a low permeate flow compared to other processes that require pressure.